Completed Projects
Previous funding has resulted in a wide range of key findings, advancing our knowledge in the tendon biomechanics or mechanobiology field. Details of these can be found here:
Tendinopathy: Treatment effects and mechanisms (Tendinopathy TEAM)
NIHR/HEE CAT Senior Clinical Lectureship scheme
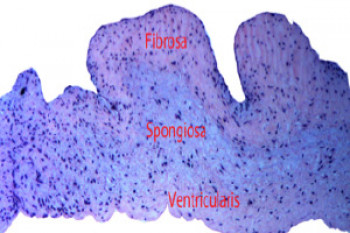
Strain transfer through the aortic valve and its relevance to the cellular mechanical environment
China Scholarship Council funded
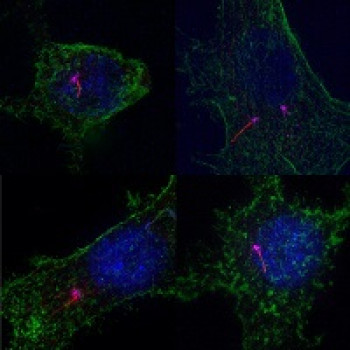
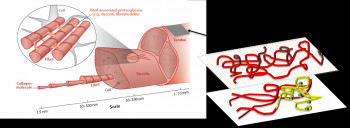
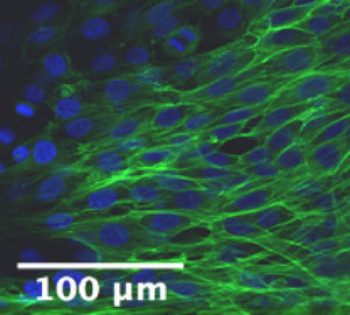
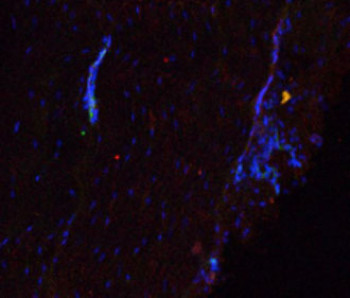
Role of interfascicular matrix in human tendons in relation to tendinopathy
Arthritis Research UK Funded: 20262
Human Tenocyte metabolism under pathological and physiological loading conditions
Arthritis Research UK Funded: NE/PhD/19598
National Institutes of Health USA Funded: R21
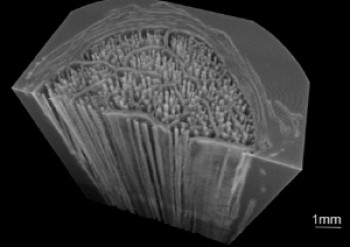
Establishing the Role of Elastin in Tendon Function
Bonfield Scholarship - Institute of Bioengineering Queen Mary University of London
Role of the inter-fascicular matrix in age related deterioration of tendon mechanical function
BBSRC Funded: BB/K008412 /1
Using hydrogels for novel tissue engineering applications
Leverhulme Trust Funded Visiting Research Professorship
A combined engineering and molecular approach to study the initiation and progression of tendinopathy
Arthritis Research UK Funded: MP 18424
A Multidisciplinary investigation into the mechanics and biology of developing tendinopathy
Wellcome Trust
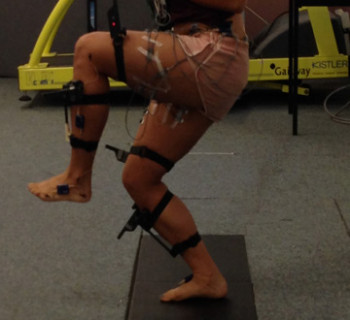
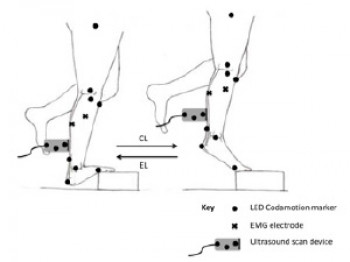
The Role of High Frequency Loading In the Treatment of Tendinopathy
Orthopaedic Research UK Funded. OR UK 490