Project
Strain transfer through the aortic valve and its relevance to the cellular mechanical environment
| Primary Investigator: | Liyong Jia |
| Co-investigator: | Hazel Screen |
| Funder: | China Scholarship Council funded |
Located between the left ventricle and aorta, the aortic valve (AV) is a cardiac tissue, serving essentially as a check valve to prevent retrograde blood flow from the aorta to the left ventricle, in each cardiac cycle. As such, it is subject to constantly fluctuating high pressure, which necessitate a robust and highly fatigue resistant tissue.
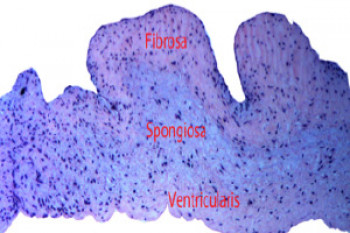
To provide fatigue resistance, the AV has a complex structure, composed of three layers, each especially optimised to meet its function. The outer fibrosa is collagen-rich to provide mechanical strength, the inner ventricularis is elastin rich for elasticity, and the spongiosa between these layers is proteoglycanaous, allowing them to shear safely, so the valve can flex back and forth during use.
The mechanical behaviour of the aortic valve is highly non-linear and it is stiffer in the circumferential than the radial direction. The aim of this project is to study how strain is transfer through the aortic heart valve under biaxial loading conditions, to see how the complex layered structure responds to loading so effectively. This understanding is critical if we are to develop future materials or tissue engineered solutions for valve repair. Further, we will assess how the resident cells in the valve - the interstitial cells – perceive the applied strain, and how they respond to it. This will allow us to investigate the possibilities for using different types of mechanical stimulus to assist in valve repair.
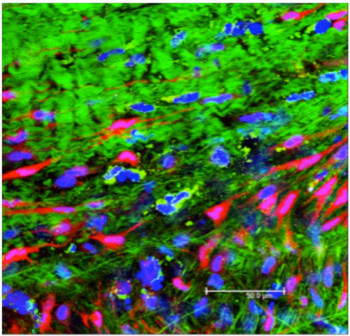
We are imaging the valve response to biaxial strain at the microscopic level using a confocal microscope, and at a macro-level using Digital Image Correlation (DIC).We are tracking cell movement and deformation with microscopy and correlating all data for a more complete understanding valve mechanics